A controlled nucleation and formation rate of self-assembled peptide nanofibers†
Abstract
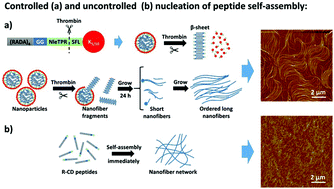
Self-assembling peptide matrixes are powerful platforms for encouraging tissue regeneration, but are usually formed within seconds and remain relatively static in both structure and function throughout their application. For the first time, we have shown that it is possible to extend the time it takes for peptide self-assembly so as to allow for the dynamic building of a self-assembled system over days, in the presence of an enzyme. Specifically, K5 and K10 sequences were conjugated, via a thrombin-specific cleavage domain NleTPR/SFL, to prevent the nanofiber formation and form stable nanoparticles composed of (RADA)4-GG-NleTPR/SFL-K5 and (RADA)4-GG-NleTPR/SFL-K10 that act as nucleation sites for reassembling. Upon introduction of thrombin, a model enzyme, this system showed an extremely slow rate of nanofiber formation in a parallel direction that is in sharp contrast to the well-known rapid assembly of (RADA)4 systems with random networks. These bioresponsive materials may provide a novel platform for utilizing long-term enzymatic profiles to form new nanofibers within an existing matrix over long therapeutic timeframes.

 Please wait while we load your content...
Please wait while we load your content...