Development of polycationic micelles as an efficient delivery system of antibiotics for overcoming the biological barriers to reverse multidrug resistance in Escherichia coli†
Abstract
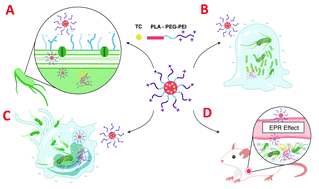
Highly pathogenic Gram-negative bacteria (G−) are tenacious and pose a serious threat to public health, mainly because of three biological barriers: cell envelope blockages, biofilm protection, and macrophages shelter. One strategy to bypass the biological barriers and consequently achieve a satisfying G− bactericidal effect is to utilize polymeric micelles with superior bacterial recognition and binding capabilities. In the current study, we explored the biological barriers penetration ability of a traditional polycationic micellar system (PP-PEI) based on a copolymer of polylactide-poly (ethylene glycol)-polyethylenimine (PLA5K-PEG2K-PEI2K). Subsequently, tetracycline (TC) with good fluorescence property was encapsulated into the PLA core of the micelle (PP-PEI/TC) through hydrophobic interaction. The combination of a PEI shell and loaded antibiotic drug endowed the polycationic micelles with a greater capacity for killing drug-resistant bacteria, destructing biofilms, and eradicating intracellular bacteria, compared with free TC and micelles without the inoculation of a PEI moiety. Confocal laser scanning microscopy (CLSM) and flow cytometry illustrated that PP-PEI/TC could completely penetrate and accumulate in drug-resistant E. coli, biofilms, and infected macrophages. The efficient biological barrier penetration was elucidated as due to the strong electrostatic interactions between the polycationic PEI block and the anionic composition of the bacterial outer membrane (e.g., LPS), macrophage cell membrane (e.g., phospholipid), and extracellular polymeric substances (e.g., eDNA), which was confirmed by biolayer interferometry (BLI). Once the micellar system was bound to a negatively-charged surface, bacterial and cellular enzymes could degrade the PP-PEI core to release its antibacterial content and finally kill planktonic bacteria, bacteria over the depth of a biofilm, and/or intracellular bacteria. In vivo imaging indicated that fluorescent polycationic micelles accumulated in bacterial infection sites with strong fluorescence. In vivo antibacterial experiments showed that PP-PEI/TC could dramatically reduce the number of drug-resistant E. coli EB1-1 in the peritoneal cavity of acute peritonitis BALB/c mice compared with its counterparts. In conclusion, our study demonstrated that polycationic micelles with a PEI shell could penetrate into drug-resistant bacteria, the biofilm matrix, and infected macrophages and lead to the spatiotemporal release of antibacterial agents for the comprehensive treatment of drug-resistant relevant infections.


 Please wait while we load your content...
Please wait while we load your content...