Unraveling the mechanism of the one-pot synthesis of exchange coupled Co-based nano-heterostructures with a high energy product†
Abstract
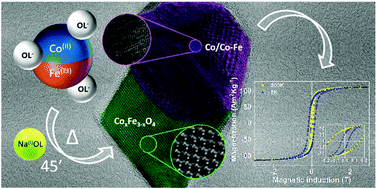
The development of reproducible protocols to synthesize hard/soft nano-heterostructures (NHSs) with tailored magnetic properties is a crucial step to define their potential application in a variety of technological areas. Thermal decomposition has proved to be an effective tool to prepare such systems, but it has been scarcely used so far for the synthesis of Co-based metal/ferrite NHSs, despite their intriguing physical properties. We found a new approach to prepare this kind of nanomaterial based on a simple one-pot thermal decomposition reaction of metal-oleate precursors in the high boiling solvent docosane. The obtained NHSs are characterized by the coexistence of Co metal and Co doped magnetite and are highly stable in an air atmosphere, thanks to the passivation of the metal with a very thin oxide layer. The investigation of the influence of the metal precursor composition (a mixed iron–cobalt oleate), of the ligands (oleic acid and sodium oleate) and of the reaction time on the chemical and structural characteristics of the final product, allowed us to rationalize the reaction pathway and to determine the role of each parameter. In particular, the use of sodium oleate is crucial to obtain a metal phase in the NHSs. In such a way, the one-pot approach proposed here allows the fine control of the synthesis, leading to the formation of stable, high performant, metal/ferrite NHSs with tailored magnetic properties. For instance, the room temperature maximum energy product was increased up to 19 kJ m−3 by tuning the Co content in the metal precursor.


 Please wait while we load your content...
Please wait while we load your content...