Co5.47N loaded N-doped carbon as an efficient bifunctional oxygen electrocatalyst for a Zn–air battery†
Abstract
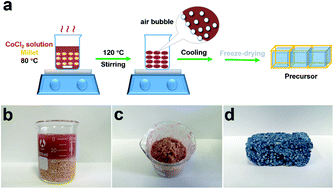
Highly active bifunctional electrocatalysts for the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) play a pivotal role in Zn–air batteries. The high cost, scarcity and instability of precious-metal-based electrocatalysts for the ORR and OER dramatically hamper their practical application in such clean-energy set-ups. Here, we report highly active Co5.47N-loaded N-doped carbon (CoNMC), prepared via the direct NH3 annealing of a millet–CoCl2 mixture, which is a cheap and mass-producible form of biomass. The optimized product shows superior ORR activity (a half-wave potential of 0.81 V vs. RHE) and electrochemical stability (a 16.5 mV negative shift of the half-wave potential after 2000 cycles) in alkaline media. Also, it shows appealing OER activity (an operating potential at 10 mA cm−2 of 1.62 V vs. RHE). This excellent electrochemical performance can be attributed to the formation of active Co5.47N nanoparticles, the large specific surface area, the abundance of nitrogen active sites, and the high graphitization degree. When assembled into a Zn–air battery, the CoNMC-based cell shows comparable performance to a Pt/C-RuO2 one.


 Please wait while we load your content...
Please wait while we load your content...