Electrochemical properties and mechanism of CoMoO4@NiWO4 core–shell nanoplates for high-performance supercapacitor electrode application studied via in situ X-ray absorption spectroscopy†
Abstract
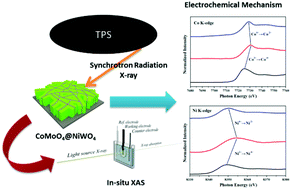
Binary transition metal oxide CoMoO4@NiWO4 core–shell nanoplates grown directly on a Ni foam substrate were synthesized via a facile two-step hydrothermal process. The core–shell nanoplates with high electrochemical surface area (2933 cm2) demonstrated excellent electrochemical properties (areal capacity as high as 0.464 mA h cm−2 at a current density of 5 mA cm−2) and great cycle stability (92.5% retention after 3000 cycles with a high current density of 40 mA cm−2). The mechanism of the electrochemical reactions based on the in situ X-ray absorption spectroscopy technique clearly shows that the Co and Ni elements simultaneously participate in the faradaic reactions with the electrolyte. These results indicate that the excellent electrochemical performance of CoMoO4@NiWO4 compared to that of CoMoO4 nanoplates is attributed to a large electrochemical surface area and synergistic effect between NiWO4 and CoMoO4. This combination of two binary transition metal oxides can hence provide an excellent route to develop a high-performance electrode material for supercapacitor applications.


 Please wait while we load your content...
Please wait while we load your content...