Enhanced charge transport via d(δ)–p(π) conjugation in Mo2-integrated single-molecule junctions†
Abstract
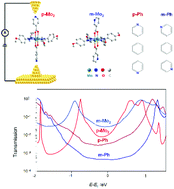
A trans-dimolybdenum nicotinate (m-Mo2) complex and its isonicotinate isomer (p-Mo2) were synthesized and characterized crystallographically, and their single-molecule charge transport properties were investigated using the STM break junction (STM-BJ) technique. With a quadruply bonded Mo2 complex unit integrated into molecular backbones, the single-molecule conductance for complex molecules was increased by more than one order of magnitude compared with that of the organic π-conjugated analogues 1,4-bis(4-pyridyl)benzene (p-Ph) and 1,4-bis(3-pyridyl)benzene (m-Ph). More interestingly, unlike m-Ph, m-Mo2 with meta connected pyridyl anchors presents larger conductance than that of p-Mo2 with two para connected pyridyl groups. DFT-based transmission calculations revealed that the significant conductance enhancement of Mo2 molecules originates from the largely reduced HOMO–LUMO gap, and the unique d(δ)–p(π) conjugation between the Mo2 unit and the pyridine rings gives rise to a delocalized electronic structure that endows the Mo2 molecules with an unexpected high conductance.


 Please wait while we load your content...
Please wait while we load your content...