Tuning the ORR activity of Pt-based Ti2CO2 MXenes by varying the atomic cluster size and doping with metals†
Abstract
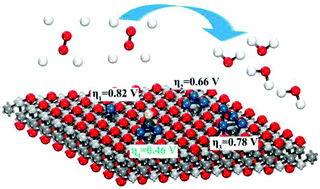
The rational design of ideal catalysts for the oxygen reduction reaction (ORR) is of great significance for solving the electrocatalytic potential problems in proton exchange membrane fuel cells (PEMFCs). Ptn (n = 1–4) and Pt3Au alloy subnanoclusters supported on a defective Ti2CO2 monolayer with oxygen vacancies (denoted as v-Ti2CO2) are simulated by using density functional theory to investigate their ORR performance. The geometries, energetics, and electronic properties of the different systems are analyzed. It is found that the supported Pt3Au alloy subnanocluster possesses the best ORR activity. The underlying mechanisms of the improved ORR activity originates from the moderate hybridization between the O 2p and the 5d orbitals of Au and Pt according to the density of states analysis. Our study suggests a facile route for designing low-cost MXene-based electrocatalysts by alloying transition metals with Pt catalysts, which may stimulate realization of suitable alternative catalysts for ORR catalysis.


 Please wait while we load your content...
Please wait while we load your content...