In situ observations of the structural dynamics of platinum–cobalt–hydroxide nanocatalysts under CO oxidation†
Abstract
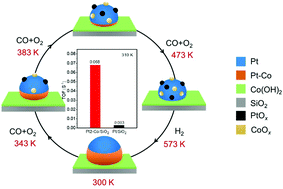
The structures, compositions and chemical states of metal catalysts are prone to dynamic changes in response to reaction conditions. In this work, a combination of in situ X-ray absorption fine structure spectroscopy and diffuse reflectance infrared Fourier transform spectroscopy has been used to monitor the temperature-dependent structural dynamics in bimetallic Pt–Co(OH)2 nanocatalysts during CO oxidation. Alloying with electron-donating Co promotes the catalytic activity of metallic Pt for CO oxidation at low temperature. At elevated temperatures under an oxidation atmosphere, O2 drives the segregation of the Pt–Co alloy into cobalt oxide and platinum metal, with the extent of alloying sharply decreasing from ∼30% at 300 K to 0 at 473 K. Reduction at high temperature could recover the formation of the Pt–Co alloy with the same alloying extent. The observed structural dynamics could be well correlated with the kinetic behavior of the catalysts. This work highlights the importance of tracking the dynamic structural changes of working catalysts for a correct understanding of their catalytic behavior.


 Please wait while we load your content...
Please wait while we load your content...