3.3 nm-sized TiO2/carbon hybrid spheres endowed with pseudocapacitance-dominated superhigh-rate Li-ion and Na-ion storage†
Abstract
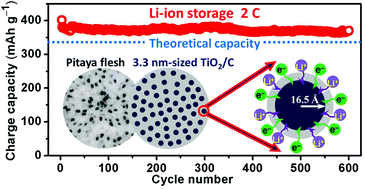
Decreasing the particle size of nanoscaled battery materials will induce amazing enhancement effects on their charging rates, which holds a promise to overcome the common bottleneck of the low charging rates of batteries. However, the fabrication of ultrafine-sized battery materials remains a great challenge. Herein, 3.3 nm-sized anatase TiO2 particles embedded in electrically and ionically conductive carbon spheres have been designed and fabricated via the suppression of Ostwald ripening with the aim to obtain insight into the electrochemical behaviors of ultrafine-sized materials. The pseudocapacitive and diffusion-controlled intercalative characteristics of the 3.3 nm-sized TiO2/carbon hybrid spheres for Li-ion and Na-ion storage have been systematically investigated via a cyclic voltammetry (CV) method combined with a differential capacitance method that is introduced here for the first time to analyze battery materials. CV and galvanostatic voltage profiles demonstrate that pseudocapacitance dominates the charge storage and increases with cycling for both Li-ion and Na-ion storage. Capacitance accounts for >83% of the Li-ion storage. A specific pseudocapacitance of 558 F g−1 with a window voltage of ∼2 V in carbonate electrolyte has been achieved. The reversible capacity is higher than the theoretical capacity of TiO2 after 600 discharge/charge cycles at 2 C and maintains ∼60% of that of TiO2 even at 80 C (45 s for full discharge or charge). For Na-ion storage, a high cycliability of 2500 discharge/charge cycles has been obtained at 2 C. Capacitance accounts for ∼79% of the Na-ion storage with cycling. Ultrafine-sized materials are very promising electrode candidates for constructing pseudocapacitive batteries possessing both high energy and power densities.


 Please wait while we load your content...
Please wait while we load your content...