Ultrastable Li-ion battery anodes by encapsulating SnS nanoparticles in sulfur-doped graphene bubble films†
Abstract
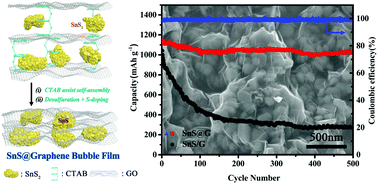
As an anode electrode material for lithium-ion batteries, SnS has high specific capacity and has received widespread attention, but its practical application is still hindered by the low reversibility of the conversion reaction and the large irreversible capacity caused by the solid electrolyte interphase (SEI). In this paper, SnS nanoparticles are encapsulated into a sulfur-doped graphene bubble film (SnS@G) by a scalable electrostatic self-assembly of SnS2/graphene oxide and hexadecyl trimethyl ammonium bromide, followed by the thermal decomposition of SnS2 and sulfur doping in graphene. Due to electrostatic attraction, the SnS nanoparticles are tightly wrapped in multilayer graphene sheets to form a flake-graphite-like structure. Compared with the disordered stacked SnS/graphene sheet composite, the closely packed SnS@G shows a much lower specific surface area and smaller irreversible Li+ consumption and surface film resistance after lithiation. The SnS@G composite anode exhibits great initial coulombic efficiency (83.2%), which is the highest value among the chemically synthesized SnS anodes. It also presents unprecedented cycling stability (1462 mA h g−1 after 200 cycles at 0.1 A g−1 and 1020 mA h g−1 after 500 cycles at 1 A g−1) and superior rate capabilities (750 mA h g−1 at 5 A g−1) upon Li storage, which demonstrates its excellent electrochemical performance and great potential as a negative electrode material for lithium-ion batteries.
- This article is part of the themed collection: Editor’s Choice: 2D Materials for Energy Storage and Conversion


 Please wait while we load your content...
Please wait while we load your content...