Naturally derived honeycomb-like N,S-codoped hierarchical porous carbon with MS2 (M = Co, Ni) decoration for high-performance Li–S battery†
Abstract
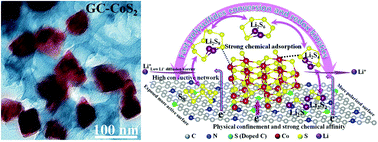
Even though lithium–sulfur batteries have appealing advantages including a high theoretical capacity and energy density, their commercial implementation has been seriously hindered by some notorious reasons, particularly the severe shuttling effect, the insulating nature of sulfur, the large volumetric variation during cycling and the sluggish redox reaction kinetics. To tackle these issues, a biomass (ginkgo-nut) derived N,S-codoped porous carbon (GC) with an interconnected honeycomb-like hierarchical structure is synthesized by a templated carbonization method, followed by hydrothermal growth of transition metal sulfide MS2 (M = Co, Ni) nanocrystals, giving rise to a hybrid 3D electrocatalyst. The unique structure constructed by N,S-codoping can expose more active sites and polar surfaces to physically confine and chemically adsorb polysulfides. Meanwhile, the embedded MS2 polyhedral-like nanoparticles further enhance the interaction with polysulfides and improve conversion and redox kinetics of polysulfides. Remarkably, with 80 wt% sulfur loading (∼2.5 mg cm−2), GC–CoS2 exhibits a reversible capacity of 988.8 mA h g−1 after 500 cycles at 0.1 C and an excellent capacity of 610.3 mA h g−1 after 1000 cycles at 2 C, outperforming bare GC and GC–NiS2. Compared with the electrochemical performances of the representative reported biomass-derived sulfur host for Li–S batteries, evidently, both the discharge capacity and cycling stability of our GC–CoS2 sample are superior. Density functional theory (DFT) calculation results suggest that CoS2 exhibits a higher binding energy towards lithium polysulfides and a lower energy barrier for Li+ diffusion on the surface compared to the NiS2 counterpart, suggesting that CoS2 is a better choice for lithium–sulfur batteries than NiS2. This work provides a new avenue to rationally design a carbonaceous catalyst host for high-performance lithium–sulfur batteries.
- This article is part of the themed collection: Nanoscale 2021 Lunar New Year Collection


 Please wait while we load your content...
Please wait while we load your content...