Controlling layered Ruddlesden–Popper perovskites via solvent additives†
Abstract
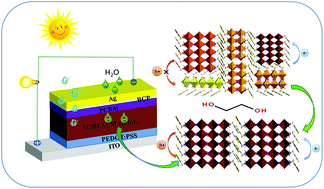
Layered Ruddlesden–Popper (RP) phase perovskites, with a formula of A′2MAn−1Pbn3n+1 (where A′ is an organic cation, MA is methylammonium, n is the integer for the number of inorganic sheets [PbI6] between the organic cation spacers), are of high interest due to their chemical stability. However, the low-n phases (e.g., n = 1, 2) not only act as carrier traps, but also hinder carrier transport within the layered RP perovskites, leading to a decreased photovoltaic performance for their corresponding devices. Herein, we report that a solvent additive-induced Ostwald ripening process effectively promotes the reduction of low-n phases and uniform RP perovskites composition. Note that the solvent added, such as ethylene glycol as an example, should have selective solubility to organic cations, and be less volatile but have a higher boiling point than the host solvents such as N,N-dimethylformamide (DMF). During fast spin-coating at room temperature, the host solvent DMF quickly evaporates while the low-n phase perovskite films are formed due to a smaller nucleation barrier, allowing the contained solvent additive, ethylene glycol, to act as a plasticizer. Then, annealing at 100 °C, causes a slow release of the restrained solvent additive which recrystallizes the perovskite grains with low-n phases into high-n phases (e.g., n ≥3) in the (ClPEA)2MA3Pb4I13 (where ClPEA is 2-(4-chlorophenyl)ethanaminium) layered RP phase perovskites. When used in solar cells, the device with a configuration of ITO/PEDOT:PSS/RP perovskite/PC61BM/BCP/Ag showed an enhanced 11% power conversion efficiency, which can be attributed to the decreased trap-state density and the increased carrier transport induced by the solvent additive.


 Please wait while we load your content...
Please wait while we load your content...