Quinone/ester-based oxygen functional group-incorporated full carbon Li-ion capacitor for enhanced performance †
Abstract
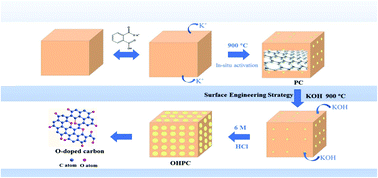
Lithium ion capacitors (LICs) are regarded as one of the most promising energy storage devices since they can bridge the gap between lithium ion batteries and supercapacitors. However, the mismatches in specific capacity, high-rate behavior, and cycling stability between the two electrodes are the most critical issues that need to be addressed, severely limiting the large energy density and long cycling life of LICs while delivering high-power density output. Herein, quinone and ester-type oxygen-modified carbon has been successfully obtained by chemical activation with alkali, which is beneficial to the absorption of PF6− together with lithium ions, which would largely improve the electrode kinetics. In particular, the cathode capacity is considerably enhanced with the increase in the amount of oxygen functional groups. Moreover, for the full carbon LIC device, an energy density of 144 W h kg−1 is exhibited at the power density of 200 W kg−1. Surprisingly, even after 10 000 cycles at 20 000 W kg−1, a capacity retention of 70.8% is successfully achieved. These remarkable results could be ascribed to the enhancement of cathode capacity and the acceleration of anode kinetics. Furthermore, the density functional theory (DFT) calculations prove that the oxygen functional groups can deliver enhanced electrochemical activity for lithium storage through surface-induced redox reactions. This elaborate study may open an avenue for resolving the issues with the electrode materials of LICs and deepen the understanding on the surface engineering strategies for incorporating oxygen-functional groups.
- This article is part of the themed collections: Nanoscale 2021 Lunar New Year Collection, 2020 Nanoscale HOT Article Collection and Nanoscale Most Popular 2020 Articles


 Please wait while we load your content...
Please wait while we load your content...