Multiscale optimization of Li-ion diffusion in solid lithium metal batteries via ion conductive metal–organic frameworks†
Abstract
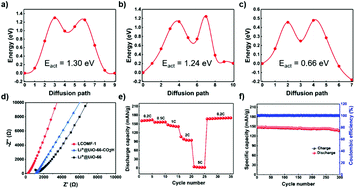
Optimization of solid electrolytes (SEs) is of great significance for lithium-based solid state batteries (SSBs). However, insufficient Li ion transport, deficient interfacial compatibility and formation of lithium dendrites lead to poor cycling performance. Based on Li+ conductive metal–organic frameworks (LCMOFs), herein a multiscale optimization strategy is put forward to facilitate Li+ transport within the MOFs (molecular scale), between the MOFs’ boundaries (nanoscale) and across the SE/electrode interface (microscale) in SSBs. LCMOFs are obtained by binding Li+ onto ionogenic chemical groups (–CO2H, –SO3H and –OH) in nanoscale dispersed MOFs. Both experimental results and DFT simulations confirm the key role of ionogenic groups for Li+ transport. Furthermore, benefiting from the optimized interfaces between LCMOF crystals, SEs with excellent electrochemical properties are obtained, including a high ionic conductivity of 1.06 × 10−3 S cm−1 at 25 °C, a wide electrochemical window from 2.0 to 4.5 V, low interfacial resistances and stable Li plating/stripping. The fabricated Li|SE|LiFePO4 SSB exhibits high and stable charge/discharge capacities under wide operation temperatures ranging from −20 to 60 °C.


 Please wait while we load your content...
Please wait while we load your content...