Structure and orientation effects in the coalescence of Au clusters
Abstract
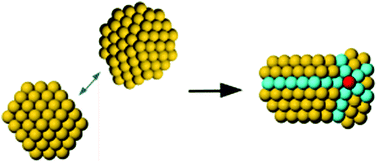
The atomic configuration of nanoparticles is key to their properties, in a way that resembles the structure–function correlation of biomolecules, and depends on the growth mechanism. Coalescence is often an important step in the growth of nanoparticles, both in liquid and in the gas phase. In coalescence, two preformed clusters collide and merge to form a larger aggregate, whose shape evolves from an initial configuration, which is strongly out of equilibrium, towards more compact structures. Here the coalescence in the gas phase of gold clusters of size ∼2 nm is simulated by the Molecular Dynamics (MD) method. Our simulations reveal a persistent influence of the structure and relative orientation of the initial colliding clusters on the final coalesced aggregates, even after more than 1 μs at temperature T = 500 K. This result is interpreted in terms of a special type of kinetic trapping, in which the choice of the structural motif takes place in the initial stages of the coalescence process. In the subsequent evolution, the coalescing aggregate may equilibrate within that motif, while transformations between different motifs are not observed, so that inter-motif equilibration is not achieved. Inter-motif equilibration is only achieved at 550 K and above. The simulations also show that aggregate reshaping occurs by a variety of specific transformation pathways, which often involve the concerted displacements of many atoms.


 Please wait while we load your content...
Please wait while we load your content...