Eliciting an immune hot tumor niche with biomimetic drug-based multi-functional nanohybrids augments immune checkpoint blockade-based breast cancer therapy†
Abstract
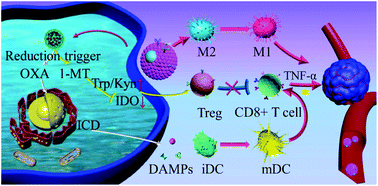
Immune checkpoint blockade (ICB) has emerged as one of the breakthrough approaches for tumor immunotherapy. However, known as an immune “cold” tumor, breast cancer harbors an immunosuppressive tumor niche that compromises ICB-based therapy. Chemoimmunotherapy combines a chemotherapeutic with an immune-modulating agent, representing a promising tactic to combat cancers, while the lack of effectively targeted co-delivery strategy is one of the main obstacles to achieve the synergistic utilization. Herein, self-assembled PEGylated pure drug-based nanohybrids (DNH) were created, which could evoke immunogenic cell death (ICD), aiding ICB-based immunotherapy by controlling the spatiotemporal release of oxaliplatin (OXA) and small molecular inhibitor 1-methyl-D-tryptophan (1-MT). Furthermore, biomimetic functionalization was exploited by nature killer cell membrane camouflaging to target cancerous cells as well as by elicit immune response through inducing M1 macrophage polarization. The drug release profiles of the nanosystem were investigated in the presence of low pH and intracellular reductants. Systemic in vivo bio-behaviors were evaluated via pharmacokinetics and biodistribution. As an “all-in-one” pure drug-based codelivery system, our biomimetic nanoplatform possessed multiple immunomodulation functions, which markedly aided in increasing the frequency of immune responders and generate an immune “hot” breast tumor niche, and eventually allowed to boost breast cancer therapy.


 Please wait while we load your content...
Please wait while we load your content...