Surface reconstruction of AgPd nanoalloy particles during the electrocatalytic formate oxidation reaction†
Abstract
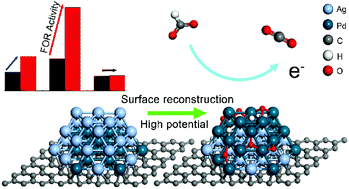
Formate is a kind of carbon-neutral fuel that can be synthesized by electrochemical conversion of CO2, however, the generated aqueous formate electrolyte is still short of potential application. Here, formate solution is proposed to be utilized as anode fuels of direct formate fuel cells through the formate oxidation reaction (FOR), and graphene supported AgPd nanoalloys (AgPd/rGO) are prepared to catalyze the FOR. Specifically, the mass activity of the as-prepared Ag49Pd51/rGO catalyst is 4.21 A mg−1Pd and the retention activity of Ag49Pd51/rGO is 49.1% of initial activity after successive 500 cycles, which is 2.48 and 3.03 times higher than that of unsupported Ag51Pd49 nanoalloys. When increasing the positive scan limit from 0.0 to 0.8 V, the mass activity of the Ag49Pd51/rGO catalyst increases from 2.32 to 6.03 A mg−1Pd and Pd surface coverage increases from 51.87% to 62.42%, indicating the occurrence of surface reconstruction where Pd atoms migrate to the surface of AgPd nanoalloys, and less intensive reconstruction is observed in unsupported Ag51Pd49 nanoalloys, whose mass activity increases from 1.35 to 2.49 A mg−1Pd. The driving force and kinetic path are calculated for the surface reconstruction induced by the adsorption of H, O and C atoms, in the case of C atoms on graphene, the segregation energy of surface Pd atoms in the AgPd nanoalloy is −1.16 eV, and the activation energy for the migration of subsurface Pd atoms to the surface is 0.54 eV, which are lower than the segregation (0.03 eV) and activation (2.06 eV) energy on a clean alloy surface.


 Please wait while we load your content...
Please wait while we load your content...