Computational screening of MN4 (M = Ti–Cu) based metal organic frameworks for CO2 reduction using the d-band centre as a descriptor†
Abstract
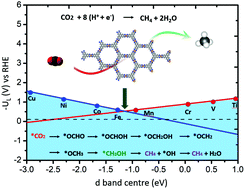
Electrocatalytic reduction is considered to be a promising way for the green and sustainable conversion of CO2 into fuels and chemicals. Transition metals, copper particularly, are the most popular catalysts for this process and a wide range of reduced carbon compounds can be obtained. In previous studies, the binding energies of *CO and *OH were adopted as descriptors to screen out the best catalyst. However, this approach is not effective for those catalysts that have a weak interaction with CO molecules. Herein, we present a theoretical work by using the d-band centre as a descriptor to predict the best catalyst for CO2 reduction to CH4 based on newly synthesized metal organic frameworks, namely porous M3 (HITP)2 (HITP, 2,3,6,7,10,11-hexaiminotriphenylene) two-dimensional metal organic frameworks (MN4-MOFs). The limiting potentials of MN4-MOFs (M = Ti to Cu) for CO2 reduction, determined by the formation energy of *OCHOH and *OCH2OH species, are closely correlated with the d-band centre from the TiN4-MOF to CuN4-MOF. Among the eight catalysts examined, the FeN4-MOF turns out to be the most active one for the selective conversion of CO2 to CH4 with an ultralow limiting potential of only −0.41 V, which is comparable or even lower than that of other reported CO2 reduction catalysts.


 Please wait while we load your content...
Please wait while we load your content...