The effect of phenylalanine ligands on the chiral-selective oxidation of glucose on Au(111)†
Abstract
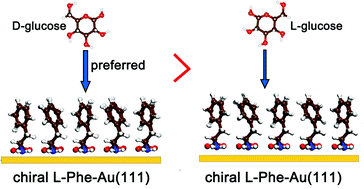
As typical glucose oxidase nanozymes, gold nanoparticles (Au NPs) have attracted much attention due to their wide-ranging applications. Ligand caps, as the “cure-all solution” for NPs, not only play important roles in the size and shape control of Au NPs but also influence their catalytic activity and selectivity. A deep understanding of the catalytic mechanism and precise description of the important role of ligands can provide possible ways to design functional Au NPs. Here, with the specific example of Au(111) capped with chiral phenylalanine (Phe), the chiral selective oxidation mechanism of glucose and the important role of the ligands were studied via first-principles calculations. All results show that the dehydrogenation of glucose to form glucono delta-lactone (GDL) is favored on clean Au(111), while the subsequent hydrolysis of GDL is the rate-limiting step for glucose oxidation. The flat and nonchiral Au(111) surface shows negligible selectivity in relation to the oxidation of D- and L-glucose, while chiral Phe-Au(111) shows selective adsorption towards D- and L-glucose. L-Phe-capped Au(111) prefers to adsorb D-glucose, while D-Phe-capped Au(111) prefers to adsorb L-glucose. Considering the three steps in the capped ligand catalysis (adsorption, replacement and reaction), we propose that the ligands play key roles in selectively adsorbing reactants before the subsequent exchange and reaction steps.
- This article is part of the themed collection: Theoretical Modelling at Nano-bio Interfaces


 Please wait while we load your content...
Please wait while we load your content...