FeNi3-modified Fe2O3/NiO/MoO2 heterogeneous nanoparticles immobilized on N, P co-doped CNT as an efficient and stable electrocatalyst for water oxidation†
Abstract
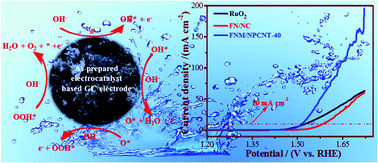
As a rate-determining step, electrocatalytic water oxidation acts a pivotal role in the water splitting process. As a consequence, it is of great significance to explore low-cost, efficient and durable electrocatalysts for the oxygen evolution reaction (OER) to promote electrocatalytic splitting water. Herein, for the first time, FeNi3-modified Fe2O3/NiO/MoO2 heterogeneous nanoparticles immobilized on N, P co-doped CNT matrix materials (FNM/NPCNT) are synthesized via a facile solid-phase grinding of the precursor, composed of nickel hexacyanoferrate/phosphomolybdic acid/CNT, and subsequently pyrolyzing under nitrogen atmosphere without any further post-processing. Due to its significant enhancement of the charge transfer efficiency and prevention of the metallic-based catalysts from being corroded, the as-prepared FNM/NPCNT hybrid electrocatalyst shows a high OER activity with a low overpotential of 282 mV vs. RHE at 10 mA cm−2 and a small Tafel slope of 46.2 mV dec−1 in an alkaline electrolyte. Moreover, the as-prepared FNM/NPCNT hybrid delivers a large mass activity of 327.6 A g−1 at the potential of 1.7 V and excellent stability (more than 20 h). This study opens up a new approach to design and synthesize non-precious transition metal-based composites immobilized N, P co-doped CNT materials as OER catalysts with high efficiency and long-term stability for promoting water splitting.


 Please wait while we load your content...
Please wait while we load your content...