Thermo- and pH-responsive fibrillization of squid suckerin A1H1 peptide†
Abstract
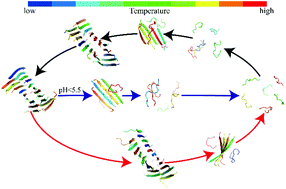
Stimuli-responsive smart materials have attracted considerable attention with numerous applications in nanotechnology, sensing, and biomedicine. Suckerin family proteins found in squid ring teeth represent such a class of peptide-based smart materials with their self-assemblies featuring excellent thermo-plasticity and pH-dependence. Similar to block copolymers, suckerin proteins are comprised of two repeating sequence motifs, where M1 motifs are abundant in alanine and histidine residues and M2 are rich in glycine. Experimental studies of suckerin assemblies suggested that M1 regions mainly formed nano-confined β-sheets within an amorphous matrix made of M2 modules stabilizing these β-rich nano-assemblies. The histidine-containing M1 modules are believed to govern the pH- and temperature-sensitive properties of suckerin assemblies. To better understand the stimuli-responsive properties of suckerin assemblies at the molecular level, we systematically studied the self-assembly dynamics of A1H1 peptides – a representative M1 sequence – at different temperatures and pH conditions with atomistic discrete molecular dynamic simulations. Our simulations with twenty A1H1 peptides demonstrated that below the transition temperature Tagg, they could readily self-assemble from isolated monomers into well-defined β-sheet nanostructures by both primary and secondary nucleation of β-sheets and subsequent aggregation growth via elongation and coagulation. Interestingly, the dissociation of pre-formed A1H1 β-sheet nanostructures featured a melting temperature Tm higher than Tagg, exhibiting the thermal hysteresis that is characteristic of first-order phase transitions with high energy barriers. In acidic environments where all histidine residues were protonated, the stability of the A1H1 β-sheet nano-assemblies was reduced and the β-rich assemblies easily dissociated into unstructured monomers at significantly lower temperatures than in the neutral solution. The computationally derived molecular mechanisms for pH- and temperature-dependent A1H1 self-assembly will help to understand the supramolecular assembly structures and functions of the large suckerin family and aid in the future design of peptide-based stimuli-responsive smart materials.
- This article is part of the themed collection: Theoretical Modelling at Nano-bio Interfaces


 Please wait while we load your content...
Please wait while we load your content...