A robust Mn@FeNi-S/graphene oxide nanocomposite as a high-efficiency catalyst for the non-enzymatic electrochemical detection of hydrogen peroxide †
Abstract
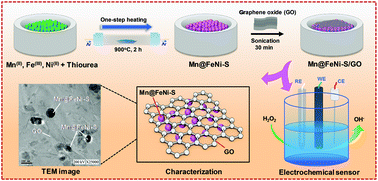
Exploring high-efficiency, stable, and cost-effective electrocatalysts for electrochemical activities is greatly desirable and challenging. Herein, a newly designed hybrid catalyst with manganese-doped FeNi-S encapsulated into graphene oxide (Mn@FeNi-S/GO) with unprecedented electrocatalytic activity was developed by simple one-step heat treatment followed by sonication. X-ray powder diffraction (XRD), Raman spectroscopy, Fourier transform infrared spectroscopy (FT-IR), field emission scanning electron microscopy (FE-SEM), high-resolution transmission electron microscopy (HR-TEM), X-ray photoelectron spectroscopy (XPS), and N2 sorption isotherm demonstrated the successful formation of Mn@FeNi-S/GO. Electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) further confirmed the kinetic-favourable adsorption of hydrogen peroxide (H2O2) onto the surface sites of Mn@FeNi-S/GO. Additionally, the synergetic effects between Mn@FeNi-S and GO are regarded as significant contributors to an efficient electron transfer path, and they promote the capture of H2O2 in hybrid catalysts. Under an optimal condition, a biosensor-based Mn@FeNi-S/GO electrode exhibits a high sensitivity of 8.929 μA μM−1 cm−2 and a detection limit of 8.84 nM with a wide detection range for H2O2 and excellent selectivity; also, it is capable of online monitoring H2O2 derived from apple juice and human blood serum.
- This article is part of the themed collection: Nanoscale 2021 Lunar New Year Collection


 Please wait while we load your content...
Please wait while we load your content...