Mechanistic modeling of spontaneous penetration of carbon nanocones into membrane vesicles†
Abstract
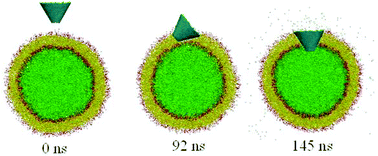
Carbon nanocones (CNCs) are promising drug delivery systems that can be functionalized with a variety of biomolecules (such as proteins, peptides, or antibodies), which allow for site-specific, targeted payload delivery to particular cells and organs. However, considerable uncertainty exists with respect to the toxicity of CNCs on their conical shape, and the underlying mechanism that leads to the penetration of CNCs (especially the truncated ones) in and through the cell membrane is not yet well understood. Using a coarse-grained dissipative particle dynamics method, we systematically investigate the spontaneous penetration of untruncated and truncated CNCs into membrane vesicles. For untruncated CNCs, the simulation results show that both pristine and oxidized ones can spontaneously penetrate across or be attached to the vesicle surface without membrane rupture, indicating low or insignificant toxicity. However, for truncated CNCs, we find that both the apex angle and aspect ratio can influence the CNC–membrane interactions and CNC-induced toxicity: a higher apex angle (and/or a lower aspect ratio) yields a higher toxicity of truncated CNCs. Further free energy analysis reveals that the lowest free energy path during the penetration is associated with CNC's orientation and rotation. For a truncated CNC with a low aspect ratio and high apex angle, it tends to rotate itself to a preferred standing-up fashion inside the vesicle membrane, posing an enhanced toxicity of CNCs. These findings may provide useful guidelines for designing effective CNC vehicles for drug delivery.
- This article is part of the themed collection: Theoretical Modelling at Nano-bio Interfaces


 Please wait while we load your content...
Please wait while we load your content...