Mechanistic insights of the reduction of gold salts in the Turkevich protocol†
Abstract
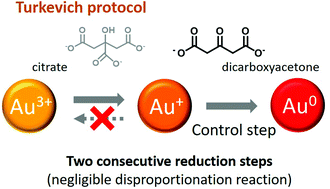
This paper presents fundamental understanding of the mechanism of the Turkevich protocol, the method recommended by the National Institute of Standards and Technology for the synthesis of gold nanoparticles using sodium citrate as reducing agent. Herein, we reveal that the Turkevich mechanism consists of two consecutive reduction steps (Au3+ → Au+ → Au0) rather than a reduction followed by the disproportionation reaction as conventionally believed. This new understanding has profound implications: i. the second reduction step (Au+ → Au0), rather than the previously postulated first reduction step, is the rate-limiting reduction step and ii. the formation of acetone dicarboxylate (DC2−) as an intermediate product through the oxidation of citrate has a key role as stabilizer and as a reducing agent (stronger than sodium citrate). This knowledge enables the synthesis of monodispersed gold nanoparticles with sizes ranging from 5.2 ± 1.7 nm to 21.4 ± 3.4 nm, with the lower end considerably smaller than previously reported through the Turkevich route. This work provides fundamental guidance for the controllable synthesis of nanoparticles using DC2− as a reducing agent directly applicable to other precious metals.
- This article is part of the themed collection: Introducing the Reaction Chemistry & Engineering Associate Editors


 Please wait while we load your content...
Please wait while we load your content...