Ideal two-dimensional solid electrolytes for fast ion transport: metal trihalides MX3 with intrinsic atomic pores†
Abstract
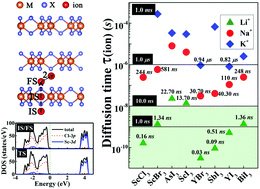
Exploring ultrathin two-dimensional (2D) solid electrolytes with fast ion transport is highly desirable in nanoelectronics, ionic devices and various energy storage systems, following the rapid scaling of devices to the nanometer scale. Herein, two-dimensional (2D) metal trihalides MX3 (ScCl3, ScBr3, AsI3, ScI3, YBr3, SbI3, YI3 and BiI3) with intrinsic atomic pore structures have been examined and found to be promising as realistic 2D solid electrolytes. Through examining the binding interactions and the diffusion barriers of monolayer MX3–ion (Li+, Na+, K+, Mg2+, and Ca2+) systems by utilizing first principles calculations, it is found that MX3–ion complexes are energetically favorable and the energy barriers of some MX3–ion systems are comparable to or even smaller than those of the conventional solid electrolyte systems. More significantly, the short diffusion time of Na+ and K+ ions in some monolayers MX3 at the nanosecond (ns) or even at the sub-ns scale indicates fast ion transport. In terms of practical applications, ultrafast Li+ travelling in the timescale of sub-ns to ns and Na+ in several tens ns in few-layer MX3 is achieved. In addition, the insulating nature of wide band gaps for MX3 is maintained during the ion transport, which is essential for solid electrolytes. These theoretical results provide fundamental guidance that MX3 materials with natural atomic pores are realistic candidates for 2D solid electrolytes with broad applications in ionic devices and energy storage devices.
- This article is part of the themed collection: Editor’s Choice: 2D Materials for Energy Storage and Conversion


 Please wait while we load your content...
Please wait while we load your content...