A nitrogen and fluorine enriched Fe/Fe3C@C oxygen reduction reaction electrocatalyst for anion/proton exchange membrane fuel cells†
Abstract
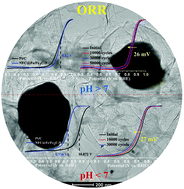
Nitrogen-doped carbon-encapsulated non-noble metals are promising electrocatalytic alternatives to Pt for the oxygen reduction reaction (ORR). Herein, we describe the efficient synthesis of nitrogen- and fluorine-doped carbon-encapsulated Fe/Fe3C (NFC@Fe/Fe3C) crystals from a Fe-poly(aniline-fluoro-aniline) co-polymer and demonstrate their use as efficient ORR electrocatalysts in acidic and alkaline environments. X-ray diffraction patterns, scanning electron microscopy, transmission electron microscopy, Raman spectra, and X-ray photoelectron spectroscopy are used to determine the structural properties of NFC@Fe/Fe3C. Of the NFC@Fe/Fe3C catalysts, NFC@Fe/Fe3C-9 demonstrates superior ORR electrocatalytic activity in both alkaline and acidic environments. NFC@Fe/Fe3C-9 follows a four-electron-transfer ORR pathway in alkaline and acidic media. Under alkaline conditions, NFC@Fe/Fe3C-9 displays a half-wave potential (E1/2) as 0.870 V, which is 16 mV higher than that of Pt/C, and its durability decay is 26 mV over 50 000 cycles. In acidic medium, the NFC@Fe/Fe3C-9 electrode shows inferior ORR performance than does Pt/C, but it is more durable, with only 27 mV decay over 30 000 cycles. A single cell performance of NFC@Fe/Fe3C-9 was tested with a proton-exchange membrane fuel cells (PEMFC) and an anion-exchange membrane fuel cell (AEMFC) with an active area of 5 cm2. The PEMFC single cell exhibits the maximum power density of 237 mW cm−2 with a back pressure of 250 kPa, while the AEMFC delivers a maximum power density of 96 mW cm−2 without back pressure.


 Please wait while we load your content...
Please wait while we load your content...