Size-dependence of the electrochemical performance of Fe–N–C catalysts for the oxygen reduction reaction and cathodes of direct methanol fuel cells†
Abstract
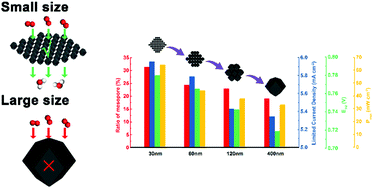
Comprehension of the structure–activity relationship is of great importance for the rational design of electrocatalysts for the oxygen reduction reaction (ORR). Herein, Fe–N–C catalysts obtained from zeolitic imidazolate framework-8 (ZIF-8) with a tunable size ranging from 30 to 400 nm are precisely synthesized. Structural investigation indicates that the catalyst with smaller size possesses a higher proportion of mesopores originating from particle stacking, which leads to enhanced catalyst utilization and accelerated mass transport. The size effect of the catalyst on ORR activity is systematically investigated by rotation disk electrode (RDE) and direct methanol fuel cell (DMFC) tests. The electrochemical performance of the Fe–N–C catalyst is found to be increased with the reduction of its particle size. The correlation among size, mesoporosity and catalyst performance is discussed, giving new inspiration for the development of rational design strategies of non-precious metal catalysts.


 Please wait while we load your content...
Please wait while we load your content...