An unexpected surfactant role of immiscible nitrogen in the structural development of silver nanoparticles: an experimental and numerical investigation†
Abstract
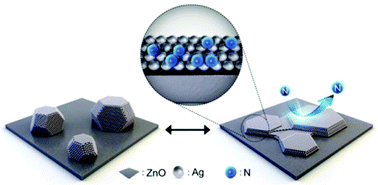
Artificially designing the crystal orientation and facets of noble metal nanoparticles is important to realize unique chemical and physical features that are very different from those of noble metals in bulk geometries. However, relative to their counterparts synthesized in wet-chemical processes, vapor-depositing noble metal nanoparticles with the desired crystallographic features while avoiding any notable impurities is quite challenging because this task requires breaking away from the thermodynamically favorable geometry of nanoparticles. We used plasma-generated N atoms as a surface-active agent, a so-called surfactant, to control the structural development of Ag nanoparticles supported on a chemically heterogeneous ZnO substrate. The N-surfactant-facilitated sputter deposition provided strong selectivity for crystalline orientation and facets, leading to a highly flattened nanoparticle shape that clearly deviated from the energetically favorable spherical polyhedra, due to the drastic decreases in the surface free energies of Ag nanoparticles in the presence of the N surfactant. The Ag nanoparticles successively developed a nearly unidirectional (111) orientation aligned by stimulating the crystalline coupling of Ag along the orientation of the ZnO substrate. The experimental and simulation results not only offer new insights into the advantages of N as a surfactant for the orientation and shape-controlled synthesis of Ag nanoparticles via sputter deposition but also provide the first solid evidence validating that immiscible, nonresidual gaseous surfactants can be used in the vapor deposition processes of noble metal nanoparticles to manipulate their surface free energies.


 Please wait while we load your content...
Please wait while we load your content...