Coupling of a conductive Ni3(2,3,6,7,10,11-hexaiminotriphenylene)2 metal–organic framework with silicon nanoparticles for use in high-capacity lithium-ion batteries†
Abstract
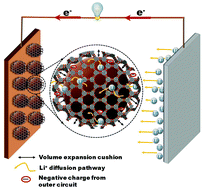
A composite of Si nanoparticles (SiNPs) and a two-dimensional (2D) porous conductive Ni3(2,3,6,7,10,11-hexaiminotriphenylene)2 (Ni3(HITP)2) metal–organic framework (MOF), namely Si/Ni3(HITP)2, is suggested as a potential anode material for Li-ion batteries (LIBs). The Ni3(HITP)2 MOF with a carbon backbone and evenly dispersed Ni and N heteroatoms showed high potential for mitigating the volume expansion of Si and enhancing the electronic conductivity as well as Li storage ability of the Si/Ni3(HITP)2 anode. The Si/Ni3(HITP)2 electrode delivered a reversible capacity of 2657 mA h g−1 after 100 cycles of discharge–charge at a rate of 0.1C. Moreover, at a high rate of 1C, the Si/Ni3(HITP)2 electrode maintained a reversible capacity of 876 mA h g−1 even after 1000 cycles. The different rate capacities were 1655, 1129, and 721 mA h g−1 at 5C, 10C and 20C, respectively. The excellent electrochemical performance of the Si/Ni3(HITP)2 electrode in terms of improved cycle life and rate capability results from the open channels of the MOF network, which are beneficial for the movement of Li+ ions through the electrolyte to the electrode and the mitigation of stress by volume expansion of Si. We believe that the coupling of conductive Ni3(HITP)2 with Si is a potential way to make an anode for high-performance LIBs.
- This article is part of the themed collection: Editor’s Choice: Functional MOFs and COFs


 Please wait while we load your content...
Please wait while we load your content...