Binding motif of ebselen in solution: chalcogen and hydrogen bonds team up†
Abstract
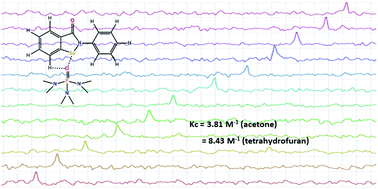
Ebselen (2-phenyl-1,2-benzoselenazol-3(2H)one), a glutathione peroxidase mimic, is active against several RNA viruses, among others the retrovirus responsible for the COVID-19 pandemic. In this paper 77Se and 1H NMR studies of ebselen are reported and they identify the chalcogen bond (ChB) and hydrogen bond (HB) that are central in the landscape of interactions formed by the compound in solution. The selenium atom and the hydrogen atom at the C7 carbon act as ChB and HB donors and the O and N atoms of neutral molecules function as acceptors. The ChB and HB give rise to a bifurcated supramolecular synthon, which fastens the interaction acceptor opposite to the N–Se covalent bond of the selenazole ring. It is known that the biologically important reaction of ebselen with cysteine thiol groups is favoured when selenium acts as a chalcogen bond donor.


 Please wait while we load your content...
Please wait while we load your content...