Birefringence and photoluminescence properties of diphenylacetylene-based liquid crystal dimers†
Abstract
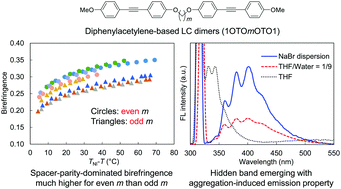
We herein report phase transitions, mesomorphism, birefringence behavior and photoluminescence properties of symmetric liquid crystal (LC) dimers based on diphenylacetylene or tolane. A homologous series of 4-methoxy tolanes linked with oligomethylene spacers consisting of carbon numbers (m) of 5–12 via ether linkages (1OTOmOTO1) were developed. The 1OTOmOTO1 series exhibited only a nematic phase, in contrast with the previously reported homologues with long terminal alkoxy chains, which exhibit layered smectic phases. We revealed that even the longest even-numbered 1OTO12OTO1 exhibits higher birefringence (Δn) than the shortest odd-numbered 1OTO5OTO1 at similar shifted temperatures. This fact suggests that the parity effect in the spacer that enhances Δn in an even-m surpasses the dilution effect that decreases Δn in large m for LC dimers. In addition, the photophysical measurements found aggregation-induced emission of the dimer concomitant with the emergence of an abnormally structured fluorescence band, which perhaps arises from a unique excited state enabled by aggregation. Single-crystal structural analysis revealed that diphenylacetylene moieties in neighboring molecules have face-to-edge orthogonal packing with one another, supporting the strong face-to-edge preferences of diphenylacetylene moiety and its enhanced fluorescence in aggregated states.


 Please wait while we load your content...
Please wait while we load your content...