Unravelling the efficient catalytic performance of ozone decomposition over nitrogen-doped manganese oxide catalysts under high humidity†
Abstract
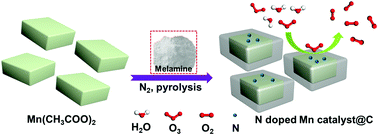
Catalytic decomposition, as a promising method for ozone elimination from ground level air or indoors, is, however, still incredibly challenging, due to the lack of stability of the available catalysts. Herein, a series of nitrogen-doped MnO-Mn2N0.86@C catalysts were prepared by pyrolysis of manganese acetate and melamine mixtures. Nitrogen-doping into the manganese oxides was observed at a high pyrolysis temperature, and this resulted in the increase of the oxygen vacancy density on the surface of the catalyst. Furthermore, the Mn species of the tubular MnO-Mn2N0.86@C-850 catalyst were coated by a carbon layer, and this could significantly improve the water vapor resistance of the catalyst under high humidity. The ozone decomposition conversion reached nearly 100% using the MnO-Mn2N0.86@C-850 catalyst, and this is clearly better than the widely studied OMS-2 catalyst and active carbon. Combined with the results of the catalytic performance and the Mn species in the catalysts, the ozone decomposition activities were revealed in the order: Mn2N0.86 > MnO2 > MnO > Mn3O4 > Mn2O3. The MnO-Mn2N0.86@C-850 catalyst showed the highest activity and stability, and this can be ascribed to the existence of a high surface area and increased oxygen vacancy density on the surface of the catalyst through nitrogen doping, as well as the presence of a hydrophobic carbon layer.


 Please wait while we load your content...
Please wait while we load your content...