p-Type Cu2O as an effective interlayer between CdS and NiOx cocatalysts to promote photocatalytic hydrogen production†
Abstract
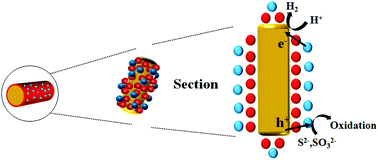
CdS has attracted considerable attention due to its suitable bandgap and excellent photocatalytic performance for producing renewable H2 energy; however, photogenerated holes in CdS move slowly relative to photogenerated electrons, which cause severe photocorrosion and a rapid surface recombination. Herein, a two-step photodeposition method is introduced, which aims to construct a p-type Cu2O interlayer between n-type CdS and NiOx cocatalyst. The photocatalyst activity with a Cu2O interlayer is nearly five times the one without the interlayer, reaching 210.86 mmol g−1 h−1. The photocatalyst with the interlayer is stable for >24 h under Xe lamp irradiation. The formation of the Cu2O interlayer is confirmed by transmission electron microscopy, X-ray photoelectron spectroscopy and energy-dispersive X-ray spectroscopy. The effect of the Cu2O interlayer is supposed to accelerate the hole transfer from CdS; thus, it promotes the charge separation and increases the H2 production activity and stability as demonstrated by electrochemical measurement. The introduction of the Cu2O interlayer and the study of its effect may yield certain guidance on the design and development of a more effective water splitting photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...