Hexyl dithiafulvalene (HDT)-substituted carbazole (CBZ) D–π–A based sensitizers for dye-sensitized solar cells†
Abstract
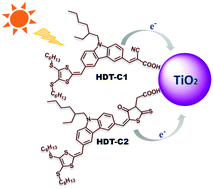
Metal-free dithiafulvalene (DTF) organic sensitizers for dye-sensitized solar cell (DSSC) applications have been greatly improved and expanded in the last decade. Herein, for DSSC applications, we modified a basic carbazole (CBZ) sensitizer (C0) by substituting additional excellent donor, hexyl dithiafulvalene (HDT), to synthesize two dithiafulvalene-carbazole (DTF-CBZ) compounds using the D–π–A concept, in which either cyanoacrylic acid (HDT-C1) or rhodanine-3-acetic acid (HDT-C2) acts as an acceptor or anchoring group. All the synthesized sensitizers were characterized by various spectroscopic and electrochemical techniques and were successfully applied in DSSCs using the I−/I3− liquid redox couple. UV-visible absorption, electrochemical studies and theoretical calculations (DFT/TDDFT) were used to determine the favoured energy levels for the sensitization of nanocrystalline TiO2. The power conversion efficiencies (PCEs) of these two dyes were compared with that of a simple carbazole sensitizer (C0); the PCE improved significantly from 3.96% (C0) to 7.38% (HDT-C1) and 6.90% (HDT-C2). The superior performance is due to the introduction of the strong electron-donating hexyl dithiafulavalene group, which leads to minimized aggregation and also favours rapid regeneration of the oxidized dye from the redox couple in DSSCs. The PCE of HDT-C2 is not drastically reduced compared to that of HDT-C1, probably due to their well-matched HOMO levels with redox potentials of the redox couple. These thermally stable DTF-CBZ organic dyes possess excellent photovoltaic properties and are anticipated to be alternatives to existing materials in DSSC applications; this will be an active methodology to fabricate DTF sensitizers in the broad and quickly growing area of metal-free organic sensitizers in solar cells.


 Please wait while we load your content...
Please wait while we load your content...