Fabrication of various morphological forms of a g-C3N4-supported MoO3 catalyst for the oxidative desulfurization of dibenzothiophene†
Abstract
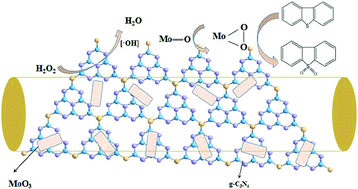
A series of catalysts consisting of nanosized MoO3 particles supported on carbon nitride with different morphologies of bulk g-C3N4 (BCN), tubulous g-C3N4 (TCN), and rod g-C3N4 (RCN) were synthesized, and the mass content of MoO3 was systematically screened. The catalysts were characterized by XRD, FT-IR, TG, SEM and TEM to understand the structure–property relationships. It proved that TCN possesses the highest pore volume compared to BCN and RCN owing to the unique morphology. MoO3 particles were highly dispersed on the surface of TCN. When the catalysts were investigated in oxidative desulfurization, MoO3/TCN gave a relatively higher desulfurization rate of 84.04%. The effects of reaction parameters on oxidative desulfurization were studied in detail, and reaction temperature, reaction time, catalyst loading, the ratio of O/S, and catalyst dosage were regarded for the optimal conditions. Under the optimal reaction conditions, of m(catalyst) = 0.013 g mL−1, T = 60 °C, O/S = 4, V(model oil) = 15 mL, and V(CH3OH) = 15 mL, the conversion of DBT could almost achieve 100%. Moreover, after six cycles of reuse, the activity of MoO3/TCN did not decrease significantly, indicating good stability. Different typical sulfur-containing substrates were selected, including BT, DBT, and 4,6-DBT. A possible mechanism for oxidative desulfurization over MoO3/TCN was proposed; hydroxyl radicals are suggested to be the main reactive species during the reaction according to the scavenger test.


 Please wait while we load your content...
Please wait while we load your content...