Au–CeO2-based nanocatalysts supported on SBA-15 for preferential oxidation of carbon monoxide (PrOx-CO)†
Abstract
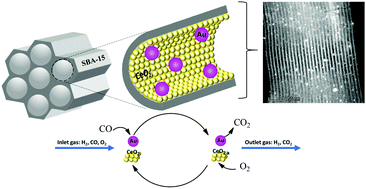
In this work, as-synthesized SBA-15 was used as a support for CeO2 and Au nanoparticles (NPs), in order to stabilize them and to promote the metal/oxide interface contact. CeO2 NPs were grown within the pores of SBA-15 using the method of successive metalorganic impregnation–decomposition cycles (IDCs), and samples with an increasing number of IDCs (x = 2, 5 and 8) were obtained. This method allowed control of the SiO2/CeO2 ratio and tailoring of samples with different crystallite sizes (2–4 nm). The (x)CeO2/S15 supports were then decorated with Au NPs through the Deposition–Precipitation (DP) method using NaOH. The DP conditions used along with the pore size limitations from each support allowed control over the Au NP size (4–9 nm). The samples were characterized by N2 physisorption, XRD, TEM, ICP-OES, XRF, DRS and Raman Spectroscopy, and for their catalytic activity for PrOx-CO. The pure supported Au-catalyst exhibited maximum CO conversion at 300 °C. The incorporation of CeO2 reduced the reaction temperature to as low as 100 °C, while the peak performance for CO conversion was observed for the catalyst containing Au and 5 IDCs of CeO2 (2.0 mol of CO2 formation per mol of Au per second at 135 °C). Further increasing the amount of CeO2 (8 IDC) resulted, however, in a decrease of the catalytic activity, resulting in a substantial reduction of the surface area and subsequent reduced access of the reaction gases (H2, CO, O2) to the active sites of the catalyst.


 Please wait while we load your content...
Please wait while we load your content...