Annulations involving 2-arylidene-1,3-indanediones: stereoselective synthesis of spiro- and fused scaffolds
Abstract
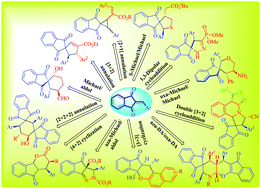
The rapid construction of three dimensional spiro- and fused cyclic skeletons is a challenging goal in synthetic organic chemistry. In this context, readily accessible 2-arylidene-1,3-indanediones meet the demand to a great extent. During the past decades, significant advances have been achieved regarding the annulation of 2-arylidene-1,3-indanediones with suitable reaction partners. This compound could serve as a multiple role player; for example, as a Michael acceptor, dienophile and diene in Diels–Alder reaction, an acceptor in 1,3-dipolar cycloaddition etc. The present review summarizes (since 2012) the stereoselective annulations involving 2-arylidene-1,3-indanediones towards the synthesis of diverse spiro- and fused carbo-/heterocyclic scaffolds based on various cycloadditions and tandem annulation methods. The applications of several important chiral organocatalysts are also highlighted. Most of the reactions described here provide quick access to the desired compounds with impressive stereoselectivity and high atom economy under mild conditions.
- This article is part of the themed collection: 2020 Focus and Perspective articles


 Please wait while we load your content...
Please wait while we load your content...