AlN4-Graphene as an efficient catalyst for CO oxidation: a DFT study
Abstract
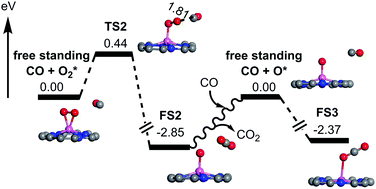
Encouraged by the previous experimental reports on the catalytic activity of aluminum porphyrins in the decomposition of water into hydrogen peroxide and hydrogen as well as the copolymerization of carbon dioxide and propylene oxide, we proposed that AlN4-Gr with the Al–N–C structure could be used as a catalyst for CO oxidation. The theoretical calculation results show that O2 is more likely to get adsorbed at the active site than CO and no energetically favorable coadsorption configurations are obtained. Combining with the difficult dissociation of O2, it is predicted that CO oxidation on AlN4-Gr first proceeds via attacking the pre-adsorbed O2 by free CO molecules to form the first CO2 with an energy barrier of 0.44 eV. The remaining atomic O is then approached by another CO molecule and finally releases CO2 without any energy barrier. The results of temperature effect further indicate that AlN4-Gr is a promising catalyst for CO oxidation at a mild temperature. It can be seen that the good catalytic activity of AlN4-Gr for CO oxidation may have originated from the moderate adsorption of O2 and O atoms. These findings offer a new route for the design of novel low-cost and efficient catalysts with main-group metals.


 Please wait while we load your content...
Please wait while we load your content...