Gamma radiation-assisted in situ synthesis of palladium nanoparticles supported on ethylenediamine-functionalized polypropylene fabric as an efficient catalyst for reduction of 4-nitrophenol
Abstract
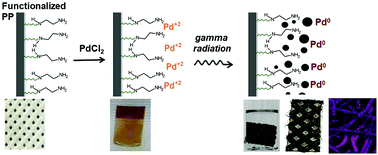
Immobilization of nanometals on a solid matrix, which offers convenient separation from the reaction mixture, is highly desirable in many chemical transformations. This work explores the use of ethylenediamine-functionalized polypropylene non-woven fabric (PP-NWF) prepared using the electron beam grafting technique as a support for the growth of PdNPs. The in situ reduction and immobilization of Pd2+ was achieved via a radiolytic route using a 60Co gamma source. The resulting Pd0 was simultaneously immobilized and stabilized through the diamino ligands present on the fabric support as confirmed by XPS and FTIR. Curve fitting of the XPS spectra obtained three characteristic Pd components identified as the surface Pd0, bulk Pd0, and Pd2+. The Pd nanoparticle (PdNP) yield was found to increase with the absorbed dose and the precursor concentration. PdNPs with average diameter sizes of 18 ± 5.4 nm, 28 ± 11 nm, and 74 ± 38 nm using 2.0 mM, 4.0 mM, and 6.0 mM initial metal ion concentrations, respectively, were observed using FE-SEM. Large PdNP clusters and a broad size distribution of the synthesized PdNPs were noted at high absorbed doses. TGA analysis showed that the synthesized Pd-EDA-f-PP-g-PGMA has good thermal stability. The use of Pd-EDA-f-PP-g-PGMA as a catalyst showed 72% conversion after 10 minutes when tested for the reduction of toxic 4-nitrophenol to 4-aminophenol, showing robust catalytic activity after 5 cycles. The present study demonstrates a facile environmentally benign approach in the preparation of supported nanoparticles as a catalyst for the degradation of an organic pollutant.


 Please wait while we load your content...
Please wait while we load your content...