The controllable nanostructure and photocatalytic behaviour of 5,10,15,20-tetra-(3,4,5 trimethoxyphenyl)porphyrin through solvophobic supramolecular self-assembly†
Abstract
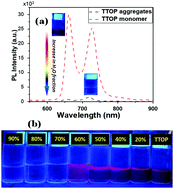
In this study, the self-assembly of 5,10,15,20-tetra(3,4,5-trimethoxyphenyl)porphyrin (coded as: TTOP) in a mixture of tetrahydrofuran (THF) and H2O and the photocatalytic activity of the resulting self-assembled aggregates toward the degradation of organic compounds were investigated. TTOP was well dispersed as monomers in THF, however, it stacked into aggregates upon the addition of water (20–90%). Depending on the THF/H2O mixture various nanostructures were observed. Microrods with an average width of 0.62 ± 0.22 μm and length of 10.17 ± 2.04 μm and sphere-like particles with diameters ranging from 0.5 to 1.2 μm were obtained in 70% water in THF. Nonetheless, the particles disappeared and the rods turned to slabs with the same length but much larger width (4.48 ± 2.27 μm) when 80% water in THF was used. Particles with irregular shapes, which connected into large aggregates, were obtained at 90% water in THF. These structures exhibited a significant difference in photocatalytic ability for the degradation of dye (rohdamine B, RhB and methylene blue, MB). The rate constants of RhB degradation were 4.35 × 10−4 min−1, 1.13 × 10−3 min−1, 2.63 × 10−3 min−1, and 3.52 × 10−3 min−1 for the porphyrin monomer in THF and its aggregate states obtained by mixing 70%, 80%, and 90% water in THF, respectively. These results indicated the important role of solvent in the formation and control of the structures of porphyrin aggregates, which are crucial to an effective porphyrin-based photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...