Catalytic performance of benzoxazine derived ordered mesoporous carbon materials for the selective oxidation of 2-methylnaphthalene
Abstract
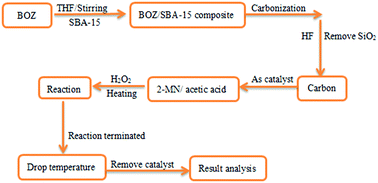
2-Methylnaphthoquinone is an important chemical product. The catalyst used in the catalytic oxidation of 2-methylnaphthalene to 2-methylnaphthoquinone has been a research hotspot. In this paper, benzoxazine derived ordered mesoporous carbon materials were used as catalysts, and their catalytic performance in acetic acid/hydrogen peroxides was investigated. The N/S co-doped carbon material (NSCM) was prepared with thiourea-type benzoxazine as a carbon precursor and SBA-15 as a template. The nitrogen-doped carbon material (NCM) was prepared using the same method using aniline-type benzoxazine as a precursor. The carbon materials were characterized by N2 adsorption/desorption, X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS) and Raman spectroscopy. The results have shown that both carbon materials exhibit worm-like structure and have graphitized amorphous structure. Nitrogen in the carbon materials existed in the form of pyridine nitrogen and pyrrole nitrogen, which caused the surface defects of carbon materials. The surface defects are critical for the enhancement of the catalytic activity of carbon materials. With NSCM and NCM as catalysts, the yields of 2-methylnaphthoquinone were 78.8% and 65.7%, respectively. By comparison, NSCM has higher catalytic activity, and the catalytic activity remains stable after five cycles of use. These results indicate that NSCM has great potential as a highly efficient catalyst for the selective oxidation of 2-methylnaphthalene.


 Please wait while we load your content...
Please wait while we load your content...