Rapid colorimetric sensing of ascorbic acid based on the excellent peroxidase-like activity of Pt deposited on ZnCo2O4 spheres†
Abstract
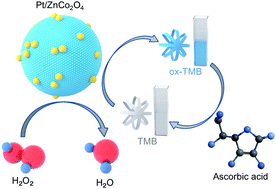
Pt deposited ZnCo2O4 nanocomposites (Pt/ZnCo2O4) were prepared by a two-step method and found to possess excellent intrinsic peroxidase-like activity. With the aid of a colorless peroxidase substrate, 3,3′,5,5′-tetramethylbenzidine (TMB), the peroxidase-like activity of Pt/ZnCo2O4 was evaluated in the presence of H2O2, which conforms to the Michaelis–Menten kinetic equation. The colorless TMB rapidly turned into a blue product (oxTMB) in just 3 min. Pt/ZnCo2O4 has a higher affinity for H2O2 and TMB compared to natural horseradish peroxidase (HRP) and can be used as a peroxidase mimic for establishing a colorimetric sensing platform. Importantly, fluorescent probe and electrochemical data verify that the catalytic activity of Pt/ZnCo2O4 is due to the accelerated electron transfer between H2O2 and TMB. Furthermore, the peroxidase-like activity of Pt/ZnCo2O4 was significantly inhibited by ascorbic acid (AA), resulting in the fading of blue oxTMB. Based on this, a simple and efficient colorimetric sensor for H2O2 and AA has been constructed. Under optimal conditions, the linear response ranges of H2O2 and AA are 100–2000 μM and 1–15 μM, respectively. The detection limits of H2O2 and AA are 26.44 μM and 0.456 μM, respectively.


 Please wait while we load your content...
Please wait while we load your content...