Magnetic BiFeO3 decorated UiO-66 as a p–n heterojunction photocatalyst for simultaneous degradation of a binary mixture of anionic and cationic dyes
Abstract
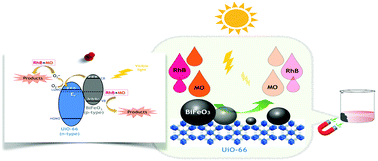
Recently, metal–organic frameworks (MOFs) have been arousing great interest as suitable materials to develop combined photocatalysts. Specifically, the superior thermal stability and unique physicochemical properties of UiO-66, a zirconium-based MOF, make it a promising candidate for the photocatalysis process. This study is the first attempt to incorporate UiO-66 with BiFeO3 by a one-pot solvothermal method for the development of visible-light photocatalysts. The prepared samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), Fourier-transform infrared spectroscopy (FT-IR), electrochemical impedance spectroscopy (EIS), thermogravimetric analysis (TGA), and diffuse reflectance spectroscopy (DRS). In this study, several UiO-66/BiFeO3 samples with various mass ratios of UiO-66 were prepared for further investigation of the influence of UiO-66 content on the photocatalytic activity of the nanocomposite. The results reveal that the UiO-66/BiFeO3 nanocomposite with 30 wt% UiO-66 shows better photocatalytic behavior for degradation of Rhodamine B (RhB), Methyl Orange (MO), and a binary dye solution (a mixture of RhB and MO) under visible-light irradiation compared to bare MOF, pure BiFeO3 and other samples. The improvement of photocatalytic performance should be attributed not only to the high surface area of UiO-66, which increases the contact area with dye, but also to the construction of a junction between UiO-66 and BiFeO3 which increases the availability of charge carriers (electrons and holes). This composing strategy of UiO-66/BiFeO3 composite may bring new insight into the design of highly efficient photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...