Selective oxidation of 1,2-propanediol to lactic acid over Cu-modified Au/hydrotalcite catalysts†
Abstract
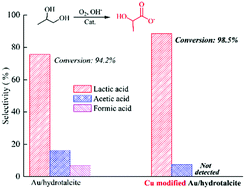
Catalytic oxidation of biomass-derived 1,2-propanediol to valuable lactic acid was investigated over various Cu-modified Au supported on Mg–Al hydrotalcite (HT). Compared to those of individual Au–HT, better catalytic performance was achieved over the Cu-modified Au–HT catalysts, mainly due to the suppression of by-product generation. The structural properties of the Cu–Au/HT catalysts were determined by XRD, SEM, HRTEM, and N2 physical adsorption, and a strong interaction was discovered in the Cu-modified Au–HT catalysts. A control experiment verified that the secondary hydroxyl in 1,2-propanediol is unreactive over Au/HT catalysts modified with Cu, which intensively inhibited the generation of the main by-products, formic acid and acetic acid. These findings provided useful guidance for the design of more efficient and stable nano-catalysts for oxidizing glycols to organic acids containing hydroxides.


 Please wait while we load your content...
Please wait while we load your content...