The metal centre in salen-acridine dyad N2O2 ligand–metal complexes modulates DNA binding and photocleavage efficiency†
Abstract
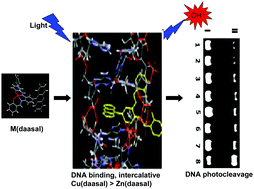
Studies on the significance of the metal centre on the activity of metal complexes have been less reported. Herein, we reported the synthesis and structural characterisation of copper and zinc complexes containing a Schiff-base type ligand, N,N′-bis(salicylidene)-9-(3,4-diaminophenyl)acridine (H2daasal). Single crystal X-diffraction analysis revealed that the structure of [Zn(daasal)] was monoclinic (space group P121/n1, unit cell parameters a = 10.6126(3) Å, b = 30.5317(10) Å, c = 11.0273(3) Å, α = 90°, β = 111.0380(10) Å, γ = 90°, V = 3334.90(17) Å3, and Z = 4) with a dihedral angle of 74° across the salophen and acridine moieties. Electron spin resonance (ESR) spectroscopy studies suggested that [Zn(daasal)] was diamagnetic and [Cu(daasal)] was paramagnetic. ESR spectral analysis revealed that the monomeric form of [Cu(daasal)] was either square planar or square pyramidal. Detailed spectroscopic analysis and viscosity studies indicate that [Cu(daasal)] binds efficiently to CT DNA (∼9 times) than [Zn(daasal)], with a slight preference for the acridine unit rather than the salen moiety. Molecular docking studies suggest that [Cu(daasal)] efficiently intercalates into DNA with higher free energy values than the [Zn(daasal)] complex. Photocleavage studies reveal that [Cu(daasal)] cleaves plasmid pBR322 DNA effectively in the presence of a reducing agent. The mechanistic studies suggest that superoxide anion radicals (O2−˙) are formed as an intermediate and proceed to produce hydroxyl radicals (OH˙) to induce DNA photocleavage. In summary, we demonstrated that changing the metal centre in an effective ligand may offer an attractive method to produce efficient DNA intercalators.


 Please wait while we load your content...
Please wait while we load your content...