Fluorescent detection of HCl in halogenated solvents via photoinduced electron transfer: towards efficient gamma radiation detection†
Abstract
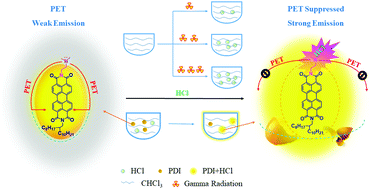
To understand the binding affinities between HCl and N,N-dimethylamine derivatives in halogen-containing solvents, and to explore the contribution of chemical substitution toward the photoinduced electron transfer (PET) process, we have designed and prepared a series of perylene tetracarboxylic diimides (PDIs) functionalized with various N,N-dimethylamine groups. All five new molecules are well soluble in chloroform, an important requirement for gamma radiation detection, and sensitive to the concentration of HCl in such a non-polar solvent. The weak fluorescence of the pristine PDIs is due to the intramolecular PET effect. After binding to HCl, the PDIs are highly fluorescent, because the PET effect is suppressed. This phenomenon is well characterized by UV-Vis spectra, fluorescence spectra, and HCl titration experiments. Their fluorescence mechanism and detection ability have been carefully studied, and their effects on the detection of gamma radiation dosage are analyzed by fitting calculations. The aromatic amine provides the lowest fluorescent background, probably because of the most efficient PET. In the cases of alkylamines, shorter chains lead to a narrower detection range, while longer chains show inefficient PET. The optimal detection limit of HCl is as low as 0.38 μM, the linear detection range for HCl is 0.38–40, and the projected linear gamma radiation detection range is 0–5 Gy, which is the widest linear detection range in comparison with the literature.


 Please wait while we load your content...
Please wait while we load your content...