Synthesis, characterization, aggregation-induced enhanced emission and solvatochromic behavior of dimethyl 4′-(diphenylamino)biphenyl-3,5-dicarboxylate: experimental and theoretical studies†
Abstract
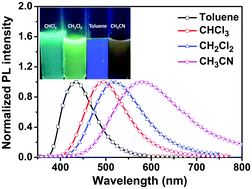
A novel triarylamine derivative dimethyl-4′-(diphenylamino)biphenyl-3,5-dicarboxylate was synthesized and characterized using spectroscopic and single crystal X-ray diffraction analysis. In addition, theoretical studies, thermogravimetric analysis, cyclic voltammetry and solvent effect studies were also investigated. As a result of solvatochromism, the molecule shows an emission depending on the solvent polarity. Increased polarity caused a red shift of the emission wavelength by 149 nm. In addition, we investigated the intramolecular charge transfer (ICT) and aggregation emission properties with respect to the packing structure of crystalline aggregates. The molecule demonstrates aggregation-induced enhanced emission (AIEE) when the water proportion is greater than 40% in an acetone/water mixture. The maximum emission intensity of the molecule was 2 times higher in an 80% water/acetone mixture. As evidenced by the crystallographic results, the phenyl ring of the compound causes steric hindrance and this hindrance prevents undesirable face to face π⋯π stacking interactions that would inevitably have an adverse effect on the fluorescence efficiency. The crystal packing of the molecule illustrates J-type stacking, which ensures that the crystal is highly fluorescent with a rigid planar structure. The results of DFT calculations are in good agreement with structural and spectroscopic data and verified the compositions of frontier molecular orbitals in similar compounds.


 Please wait while we load your content...
Please wait while we load your content...