Chiral liquid chromatography-mass spectrometry (LC-MS/MS) method development with β-cyclodextrin (β-CD) derivatized chiral stationary phase for the enhanced separation and determination of flurbiprofen enantiomers: application to a stereoselective pharmacokinetic study†
Abstract
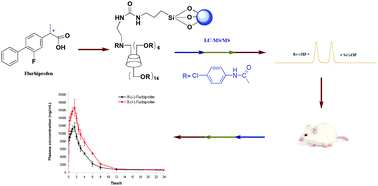
A method was developed and validated for the enantioselective determination of flurbiprofen in rat plasma using liquid chromatography/electrospray ionization-tandem mass spectrometry under reversed-phase elution mode. Two polysaccharide derivatized chiral stationary phases and a homemade β-cyclodextrin (β-CD) derivatized based chiral column were evaluated. The latter one, a per-4-chlorophenylcarbamate-β-cyclodextrin bonded chiral stationary phase which was synthesized in our laboratory, enabled the highly sensitive detection and complete separation (resolution 2.0) of the flurbiprofen enantiomers. The assay was carried out after the solid-phase extraction procedure with C18 cartridges, and with R-(−)-ibuprofen used as the internal standard. The developed method has been validated for specificity, linearity, accuracy, precision, recovery, matrix effect, stability, carryover effect and dilution effect. The lower limit of quantification for R-flurbiprofen and S-flurbiprofen was 10 ng mL−1 in rat plasma, respectively. Linearity was confirmed in the range of 10.0–20000.0 ng mL−1 with a correlation coefficient (r2) greater than 0.996. The established method was successfully applied to a stereoselective pharmacokinetic study of flurbiprofen enantiomers in rat plasma following oral administration.


 Please wait while we load your content...
Please wait while we load your content...