Energetic N-azidomethyl derivatives of polynitro hexaazaisowurtzitanes series: CL-20 analogues having the highest enthalpy†
Abstract
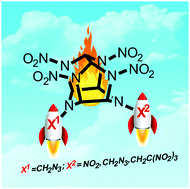
High endothermicity is one of the most important factors in the design of high-performance energetic materials for a new generation of rocket technologies. Taking this into account, in this study, novel highly energetic cage compounds were synthesized by the incorporation of a high-enthalpy N-azidomethyl moiety into the polynitro hexaazaisowurtzitane backbone. All obtained compounds were fully characterized by high-resolution mass spectrometry, IR and multinuclear NMR (1H, 13C, 14N, 15N) spectroscopy. For two of them, 4-azidomethyl-2,6,8,10,12-pentanitrohexaazaisowurtzitane and 4,10-bis(azidomethyl)-2,6,8,12-tetranitrohexaazaisowurtzitane, the density and structural features were established by X-ray diffractometry. Enthalpies of formation were determined experimentally by combustion calorimetry. Thermal stability measurements and testing of impact and friction sensitivity were also carried out. The synthesized compounds exhibit a high level of heat resistance (decomposition onset 180–205 °C) and density (1.85–1.93 g cm−3). Their enthalpies of formation on a unit of mass (up to 2210 kJ kg−1) are the highest among the known hexaazaisowurtzitane derivatives and are 2–2.5 times that of CL-20. The combination of these attractive properties makes the new materials promising energetic components for propellant compositions.


 Please wait while we load your content...
Please wait while we load your content...