Synchronous role of coupled adsorption and photocatalytic oxidation on the hybrid nanomaterials of pectin and nickel ferrite leads to the excellent removal of toxic dye effluents†
Abstract
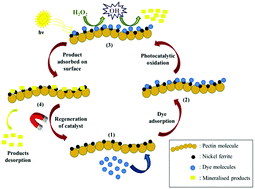
Hybrid nanomaterials of pectin and nickel ferrite were fabricated and served as potential environmentally benign materials for the treatment of toxic pollutants. Pectin and nickel ferrite mutually link and coexist to form nanocomposites, which possess coupled performance of adsorption and photocatalytic oxidation. Various characterization techniques, such as powder XRD, FT-IR, VSM, FE-SEM, HR-TEM and DRS, were employed to systematically elucidate the structural and surface properties of the synthesized nanocomposites. The influence of varying the weight% of pectin on the nanocomposites was also investigated in order to perceive the effects of the change of concentration on the adsorptive degradation efficiency. The rate constant value for the removal of methylene blue and remazol black using pectin–nickel ferrite (5 : 1) nanocomposite was 14.7 × 10−2 min−1 and 10.9 × 10−2 min−1, respectively, while it was calculated to be 2.75 × 10−2 min−1 and 1.22 × 10−2 min−1, respectively, when nickel ferrite was used. Thus the nanocomposites exhibited much enhanced removal efficiency compared to the bare nickel ferrite nanoparticles. The results elucidated that the synchronous role of coupled adsorption and photocatalytic oxidation leads to excellent removal efficiency by the nanocomposites. The inherent magnetic character of nickel ferrite facilitates the simplistic external magnet aided recovery of the hybrid nanomaterial. It is anticipated that the designed hybrid nanomaterial will serve as an eco-friendly, economically viable, highly efficient and reusable catalyst for the removal of toxic organic pollutants.


 Please wait while we load your content...
Please wait while we load your content...